With respect to the medical device industry (FDA’s QSR and standards such as ISO 13485), the team members are required to be trained in the QMS tasks that they perform as a foundation to Agile. Collective ownershipĬollective ownership involves the team taking ownership, assuming accountability and responsibility for the quality of the delivered software product. In summary, retrospectives and reflections enable ownership of continuous process improvement. It is recommended that these practices be performed regularly and frequently during the completion of each iteration, thus creating a positive atmosphere for the team to practice self-awareness inasmuch as attempting to shorten the software development cycle while maintaining effectiveness and providing quality outputs to increase user friendliness. The essential question is "How is the team currently working to complete the overall project?" Retrospectives are event-based (e.g., at the end of an iteration, a release of a product, or any significant milestone) and are part of what the learned approach encompasses (i.e., what happened during this event that will help us improve?), whereas reflections are performed periodically, depending on the nature of the team and the circumstances during a specific timeframe. Retrospectives and reflections are practices that enable the team to regularly inspect their performance and take proactive actions to increase their overall effectiveness. These can pinpoint excursions from the initial intended use of the device by identifying abnormalities in the form of a punch list or other traceable means known/used by the stakeholders. 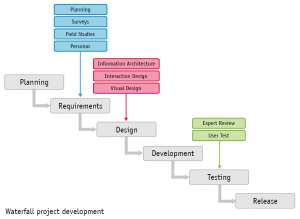
In addition, these monitoring tools can be used as mechanisms to identify significant process issues in a formal CAPA (corrective and preventive action), as required by medical device regulations per FDA QSR 21 820.100, Subpart J, and ISO 13485, Clause 8.5. 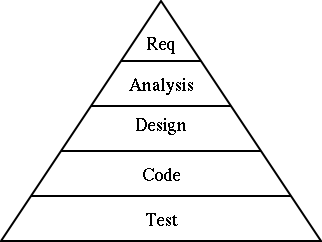
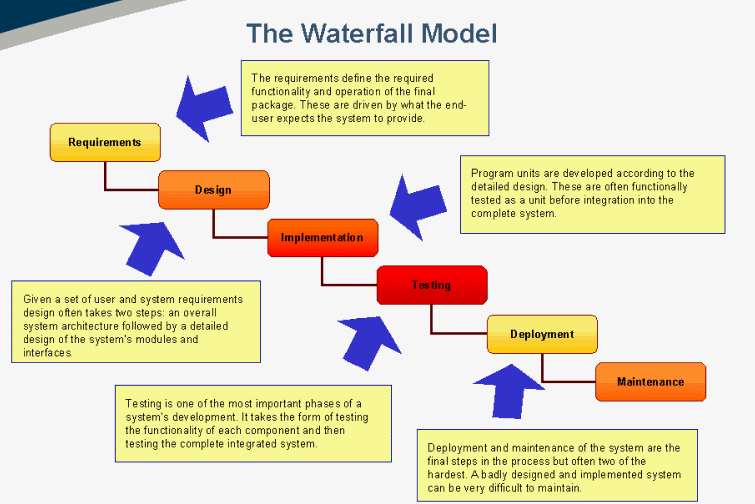
These can include burndown charts, information transparency for all applicable stakeholders, acting on the input information provided to resolve the anomaly, and creating a positive work culture where alterations are welcomed. The notion of “stop the line” involves creating tools that allow the stakeholders to monitor processes or quality gates during the development of the software. Instead, additional peers or cross-functional teams may be required to ensure that all the angles are satisfied with respect to the scope of work mandated by the customer in terms of the level of risk, while maintaining alignment with the applicable regulations. However, in cases involving more complex software development and more demanding industry expectations, pairing may be inadequate. Pairing two open-minded technical people who have different, independent perspectives means they can brainstorm their ideas while simultaneously adding value to the mission of developing the software throughout its life cycle. The ideal team structure requires hiring the right team members who understand the Agile approach while continuously collaborating throughout the software development cycle. This can also be applied within the pharmaceutical, biopharmaceutical, cell & gene therapy, and vaccine industries. Configuration management and change managementĪttempting to align Agile practices with regulatory goals, concepts, and practices entails accounting for the following elements prior to introducing any Agile approaches into your medical device quality management system (QMS).Rationale for software system testing phase.In part 3 of this series, we will dive into the following topics: Integration and integration testing phase.Implementation and unit verification phase.Defining requirements and product definition phase.In this article, which is part 2 of three, we will dive further into the use of Agile methodology, including a discussion of practices toward alignment among the pharmaceutical, biopharmaceutical, vaccine, medical device, and cell/gene therapy industries. Part 1 of this article series dived into the use of Agile methodology with a focus on the planning phase of a software development plan. The pharmaceutical industry, under the FDA and other regulatory bodies, has been utilizing computer software/systems validation testing methodology to validate software under GxP environment in alignment with the principles of good automated manufacturing practices (GAMP) since the first published guidance in 1994. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
